Authors:
Quesnay, Jose Edwin Neciosup [1, 2] ; Pollock, Naomi L. [3] ; Nagampalli, Raghavendra Sashi Krishna [1, 4] ; Lee, Sarah C. [3] ; Balakrishnan, Vijayakumar [5,6] ; Dias, Sandra Martha Gomes [1] ; Moraes, Isabel [7,8] ; Dafforn, Tim R. [3] ; Ambrosio, Andre Luis Berteli [5]
Abstract:
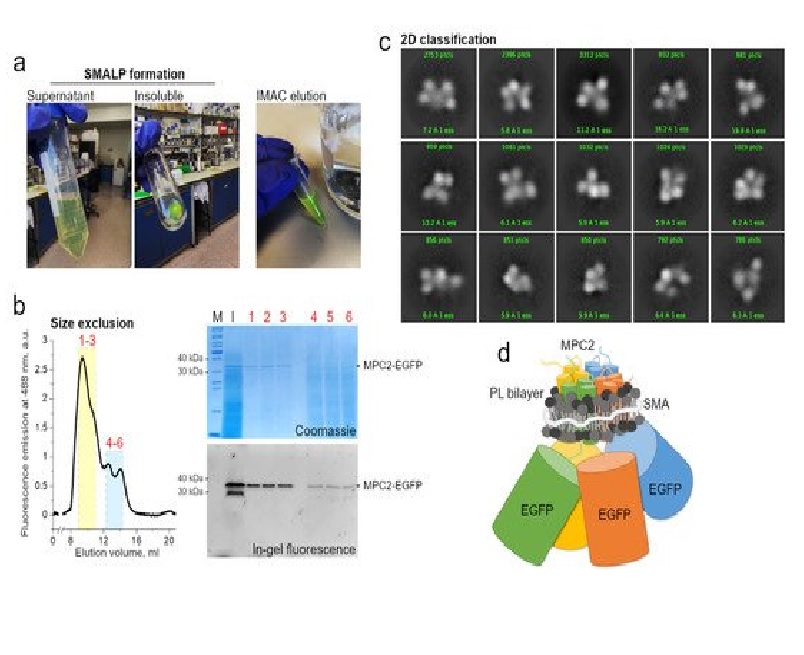
Simple Summary The atomic structure of a biological macromolecule determines its function. Discovering how one or more amino acid chains fold and interact to form a protein complex is critical, from understanding the most fundamental cellular processes to developing new therapies. However, this is far from a straightforward task, especially when studying a membrane protein. The functional link between the oligomeric state and complex composition of the proteins involved in the active mitochondrial transport of cytosolic pyruvate is a decades-old question but remains urgent. We present a brief historical review beginning with the identification of the so-called mitochondrial pyruvate carrier (MPC) proteins, followed by a rigorous conceptual analysis of technical approaches in more recent biochemical studies that seek to isolate and reconstitute the functional MPC complex(es) in vitro. We correlate these studies with early kinetic observations and current experimental and computational knowledge to assess their main contributions, identify gaps, resolve ambiguities, and better define the research goal. The molecular identity of the mitochondrial pyruvate carrier (MPC) was presented in 2012, forty years after the active transport of cytosolic pyruvate into the mitochondrial matrix was first demonstrated. An impressive amount of in vivo and in vitro studies has since revealed an unexpected interplay between one, two, or even three protein subunits defining different functional MPC assemblies in a metabolic-specific context. These have clear implications in cell homeostasis and disease, and on the development of future therapies. Despite intensive efforts by different research groups using state-of-the-art computational tools and experimental techniques, MPCs’ structure-based mechanism remains elusive. Here, we review the current state of knowledge concerning MPCs’ molecular structures by examining both earlier and recent studies and presenting novel data to identify the regulatory, structural, and core transport activities to each of the known MPC subunits. We also discuss the potential application of cryogenic electron microscopy (cryo-EM) studies of MPC reconstituted into nanodiscs of synthetic copolymers for solving human MPC2.
1 Brazilian Biosciences National Laboratory, Center for Research in Energy and Materials, Campinas 13083-970, Brazil
2 Postgraduate program in Biosciences and Technology of Bioactive Products, Institute of Biology, University of Campinas, Campinas 13083-970, Brazil
3 School of Biosciences, University of Birmingham, Birmingham B15 2TT, UK
4 Infection and Immunity Program and Department of Biochemistry and Molecular Biology, Biomedicine Discovery Institute, Monash University, Clayton 3800, Australia.
5 São Carlos Institute of Physics, University of São Paulo, São Carlos 13563-120, Brazil
6 McArdle Laboratory for Cancer Research, Department of Oncology, School of Medicine and Public Health, University of Wisconsin at Madison, Madison, WI 53705, USA.
7 National Physical Laboratory, Teddington, Middlesex TW11 0LW, UK
8 Membrane Protein Laboratory, Diamond Light Source Limited, Harwell Science and Innovation Campus, Didcot OX11 0DE, UK
Link to article: https://www.mdpi.com/2079-7737/9/11/407