Authors:
de Oliveira, Valdeir C. [1] ; de Oliveira, Juliana M. [1] ; Menezes da Silva, Vitor H. [1] ; Khan, Ismat U. [2] ; Correia, Carlos Roque D. [1]
Abstract:
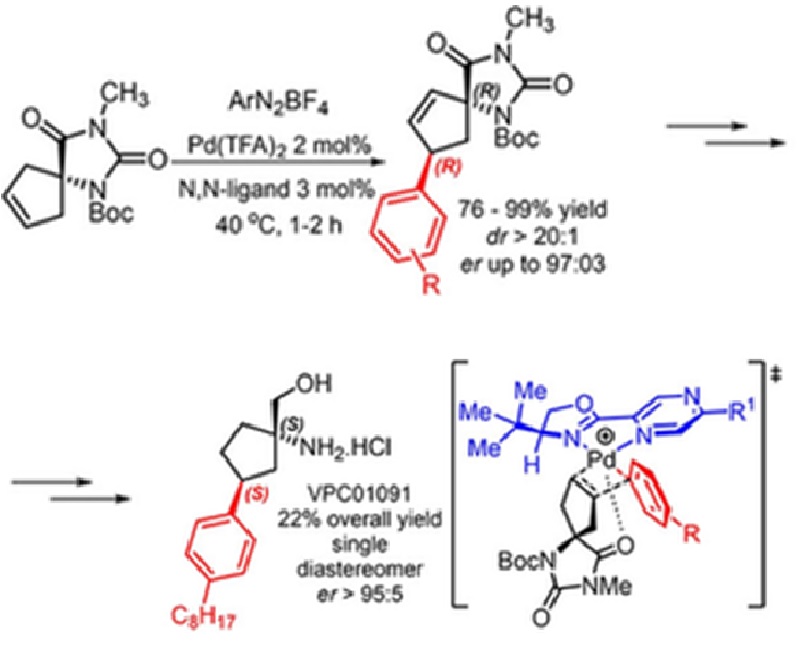
A highly efficient Heck-Matsuda desymmetrization of unsaturated spirohydantoins directed by non-covalent interactions, which allows the construction of two simultaneous stereogenic centers, including a trisubstituted quaternary one, is described. This Heck arylation permitted a novel enantioselective total synthesis of the S1PR1 agonist (also an S1PR3 antagonist) compound VPC01091, a potential drug for the treatment of multiple sclerosis. The broad scope of these enantioselective Heck-Matsuda protocol provided several arylated spiro systems in yields ranging from 76% to 99%, with enantiomeric ratios (er) up to 97:3, and diastereoselectivities (dr) of > 20:1 in all cases studied. The method uses only 2% of Pd(TFA) 2 and 3 mol% of the chiral N,N-ligand Pyrabox in short reaction times of 1-2 h. These enantioselective Heck arylations can also be carried out at the gram scale in high yields with no erosion of their diastereoselectivity or enantioselectivity. The key spiro Heck products (R,R)-21 and (R,R)-27 bearing an aryl iodide moiety or an aryl n-octyl moiety were employed as starting materials for the total enantioselective syntheses of the (S,S)-VPC01091, in overall yields of 20% and 22% respectively after 10 or 9 steps from the starting spirohydantoin, with an er> 95:5. Computational analysis of the enantioselective Heck-Matsuda desymmetrization supports the rationale involving a key non-covalent interaction between the imide carbonyl of the spirohydantoin and the cationic palladium bounded to the chiral N,N-ligand.
1 Departamento de Química Orgânica, Instituto de Química, Universidade Estadual de Campinas, CP 6154, 13083-970 Campinas-SP, Brazil
2 Institute of Chemical Sciences (ICS), Gomal University, Dera Ismail Khan, 29220 Pakistan
Link to article: https://onlinelibrary.wiley.com/doi/abs/10.1002/adsc.202000443