Authors:
Philadelpho, Biane Oliveira 1 ; Novais da Cunha, Mariana Mota 1 ; Reis Batista, Fabiana Pacheco 1 ; da Silva, Jaff Ribeiro 1 ; Druzian, Janice Izabel 1 ; Castilho, Marcelo Santos 1 ; Cilli, Eduardo Maffud 2 ; Ferreira, Ederlan S. 1
Abstract:
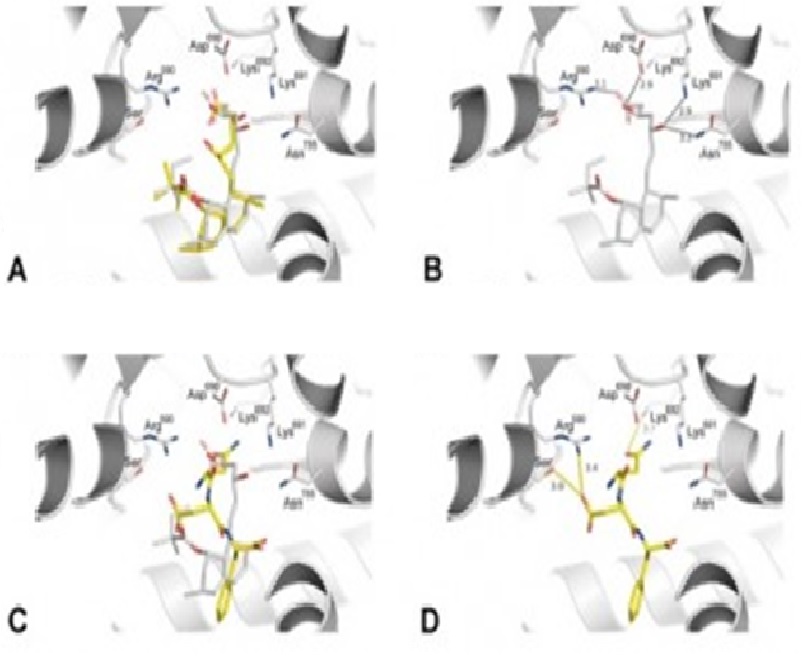
Previous studies have shown that cowpea protein positively interferes with cholesterol metabolism. In this study, we evaluated the ability of the fraction containing peptides of <3 kDa, as well as that of the Gln-Asp-Phe (QDF) peptide, derived from cowpea β-vignin protein, to inhibit HMG-CoA reductase activity. We established isolation and chromatography procedures to effectively obtain the protein with a purity above 95%. In silico predictions were performed to identify peptide sequences capable of interacting with HMG-CoA reductase. In vitro experiments showed that the fraction containing peptides of <3 kDa displayed inhibition of HMG-CoA reductase activity. The tripeptide QDF inhibits HMG-CoA reductase (IC50 = 12.8 μM) in a dose-dependent manner. Furthermore, in silico studies revealed the binding profile of the QDF peptide and hinted at the molecular interactions that are responsible for its activity. Therefore, this study shows, for the first time, a peptide from cowpea β-vignin protein that inhibits HMG-CoA reductase and the chemical modifications that should be investigated to evaluate its binding profile.
1 School of Pharmacy, Federal University of Bahia, Barão de Jeremoabo Street, 40170-115 Salvador, BA, Brazil
2 Institute of Chemistry, Sao Paulo State University (UNESP), Rua Prof. Francisco Degni, 14800-0600 Araraquara, SP, Brazil
Link para o artigo completo:
https://www.sciencedirect.com/science/article/pii/S0308814618305740?via%3Dihub